Kimberly R. Daley, Kevin J. Kubarych J. Phys. Chem. B (2017) 121, 10574-10582
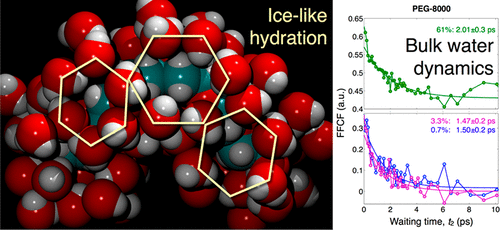
Ultrafast picosecond time scale two-dimensional infrared (2D-IR) spectroscopy of a new water-soluble transition metal complex acting as a vibrational probe shows that over a range of concentration and poly(ethylene glycol) (PEG) molecular mass (2000, 8000, and 20000 Da) the time scale of the sensed hydration dynamics differs negligibly from bulk water (D2O). PEG is well-known to establish a highly stable hydration shell because the spacing between adjacent ethereal oxygens nearly matches water’s hydrogen-bonding network. Although these first-shell water molecules are likely significantly retarded, they present an interface to subsequent hydration shells and thus diminish the largely entropic perturbation to water’s orientational dynamics. In addition to the longer PEGs, a series of concentration-dependent 2D-IR measurements using aqueous PEG-400 show a pronounced hydration slowdown in the vicinity of the critical overlap concentration (c*). Comparison between these dynamical results and previously reported steady-state infrared spectroscopy of aqueous PEG-1000 solutions reveals a strikingly identical dependence on number of water molecules per ethylene oxide monomer, scaled according to the critical overlap concentration.